

Squalene peroxide is associated with initiation of inflammatory acne cascade, triggering cytokines and lipoxygenase pathway [6]. In addition squalene peroxides diminish the important skin antioxidant glutathione, while pre-treatment with glutathione depleting agents (DL-buthionine sulfoximine and dethyl maleaate) makes the comedogenic potential of squalene peroxides even worse [7]. Accumulation of lipid peroxide in comedones caused by oxidation of sebum is associated with progression of acne vulgaris and is involved in development of acne [8].
Once inflammatory reactions initiated, more generation of, reactive oxygen species, ROS is expected [9]. ROS and free radical oxidative damage is well recognized cause of skin aging and inexorably enhance our attention toward reinventing the way we conceive anti aging acne treatment as this inevitable process affects more individuals suffering from acne vulgaris. Adult acne as opposed to adolescent acne, more common in male, occurs more frequently in female and often extends to age beyond thirty. Therefore, aging process consideration turns out to be an essential parameter to be cognizant of in treatment of this group of patients.
Presence of inflammatory factors is evident in acne vulgaris and well-known as one of the cornerstones in pathogeneis of inflammatory acne lesions[10]. However, mechanisms promoting inflammation has been less studied. Keratinocytes exposed to P. acnes increase production of reactive oxygen radicals(ROS) in particular superoxide. ROS are handled by glutathione and SOD system to produce GSH-x and finally water, which explains why SOD and GSH-Px are exhausted in acne patients [11]. Drugs with known comedolytic activities such as retinoic acid derivaties show significant inhibition of the p. acnes-stimulated superoxide production , IL-8 release and keratinocyte inhibition [12].
Not only in acne vulgaris but as well in acne inversa, hidradenitis suppurative, the patterns of inflammation have been observed, evidenced by habits of proinflammatory diets, high in saturated fatty acids, trans fats and low in complex carbohydratess and omega-6 fatty acids and more ameliorated outcomes with dietary modifications.
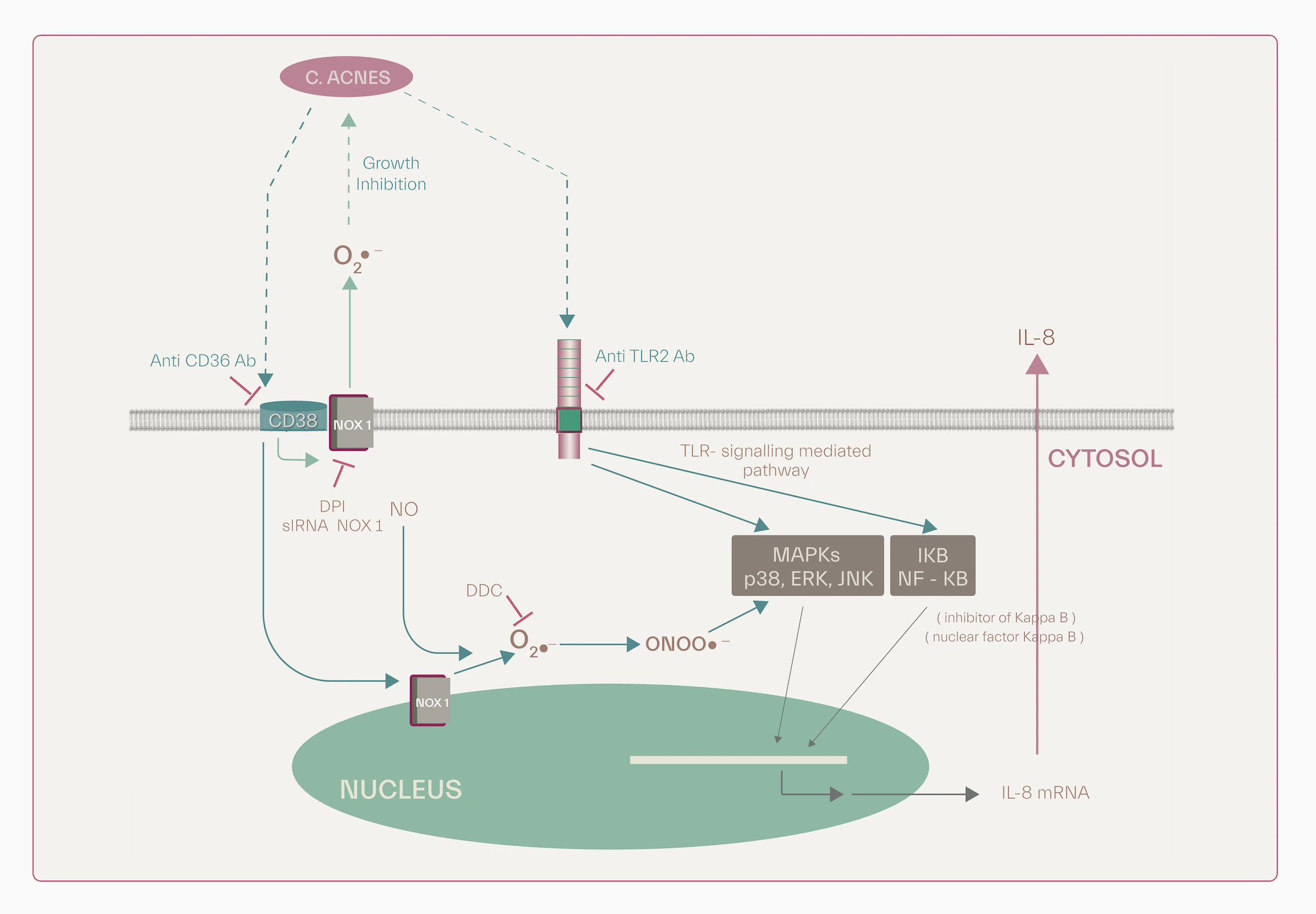
Blood antioxidant enzyme activities including superoxide dismutase (SOD) and gluttathione peroxidase (GSH-Px) in patients with papulopustular types of inflammatory acne are significantly lower than that of controls [12]. Low blood SOD, GSH-Px and malondialdehyde (MDA), a marker of oxidative stress, are characteristic of acne patients [13]. CD36 and TLR-2 recognize P.acnes. CD36 triggers the production of peroxide through NADPH oxidase pathway (NOX) and combines with NO to form peroxinitrites which, in turn, activate p38 and ERK MAPKs, thus contributing to IL-8 production. TLR-2 signaling pathway provokes IL-8 generation [11].
Some studies suggest therapeutic effects of antibiotics with antioxidants activity for inflammatory acne treatment without exerting a bactericidal/bacteriostatic effect against acne bacteria. Minocyclin and other tetracyclines, known to have clear antioxidant activities [14], at sub-antimicrobial doses, reduced papules and pustules by over 80% after 3 months in those with moderate acne [15]. However, growing tetracycline resistance, acquired through 16S rRNA mutation, [14-3], observed in 9.2% of skin samples in one study [14-1], and adverse effect during pregnancy, tooth discoloration in infants whose mothers took the drug during pregnancy [14-2], limits their pragmatic use in clinical practice.
Another aspect of lipid peroxidation in pathogenesis of acne vulgaris is from studies which suggest association between inflammatory markers, such as IL-1, IL-17 and IL-22, and upregulation of antimicrobial proteins, such as psoriasin, calprotectin, RNase 7, and human beta defensin, hBD-2 and hBD-3 [16]. Such a scenario has been reported for Propionibacterium acnes, an abundant skin commensal, preferring lipid-rich habitats such as sebaceous glands. P.acnes produces a lipase which is able to generate free fatty acids through degradation of skin lipids. This leads to an accumulation of free fatty acids such as oleic acid which promote skin adherence and colonization of P.acnes, hyperproliferation and inflammation [17].
Antioxidants has been widely used in treatment of skin disease for their protective effect against free radical species. Reactive oxygen species, notorious for their tissue injury and auto oxidative damage by attacking DNA or membrane lipids, are generated as a result of neutrophil chemotaxis and inflammatory reactions in response to propinibacterium.acnes colonization [18]. Thin layer chromatography of skin surface lipids have been demonstrated of having higher level of polar lipids, which are comedogenic by-product of squalene, a major constituent of sebum with six double bonds, high oxygen scavenging capability and high susceptibility to oxidation. Moreover, antioxidants may be potentially beneficial by preventing infiltrated neutrophils from releasing reactive oxygen species and restraining concurrent inflammation [20].
One of the most well-known antioxidants used in skin disorders is ascorbic acid [19]. Sodium L-ascorbyl phosphate, SAP, has been demonstrated effective in treatment of acne vulgaris [20]. Additionally one study indicates synergism of this compound with retinol [21]. Another study has been suggested that 1% sodicum ascorbyl phosphate (SAP) has a a strong antimicrobial effect with a log reduction of 5 after 8 hours on P. acnes in a time-kill study [22]. Further, on in a human in vivo study with 20 subjects an SAP O/W formulation significantly prevents the UVA-induced sebum oxidation up to 40% [22]. Another study indicates superiority of sodium L-asorbyl 2-phosphate compare to 1% clindamycin gel in treatment of acne vulgaris [23].
Several factors influence production of sebaceous glands but it is predominantly hormonally stimulated. Androgens especially from the testes and adrenals, stimulate the sebaceous gland directly and influence acne inflammations, whereas terminal sebocyte differentiation is assisted by peroxisome proliferator-activated receptro (PPAR) ligands [24]. Besides, sebaceous glands are important formation site of active androgens. L-ascorbic acid 2-phosphate has been suggested for its inhibition of DHT (dihydrotestosterone) by repressing DHT-induced DKK-1 (dickkopf-1) protein expression as examined by enzyme-linked immunoabsorbent assay (ELISA) [25]. Furthermore, inflammation is another primary event in acne vulgaris which may be curbed by constant delivery of vitamin C to the skin cells through topical application of magnesium ascorbyl phosphate , which explains antioxidants may counteract ROS through two principal mechanisms [41].
Zinc treatments
Zinc ascorbates is among new suggested modalities supported by one study done in Japan. In this study zinc ascorbate has been shown to have superoxide dismutase-like activity [26]. Superoxide dismutase, SOD, is an enzyme that controls reactive oxygen species production. This study substantiates a role for zinc ascorbate for treatment of acne vulgaris with its antimicrobial activity especially against staph aureus and E.coli. This antimicrobial activity is significantly higher than that of either zinc or ascorbic acid alone even in a lower concentration. Zinc treatments to ethanol-intoxicated rats was able to bring down the elevated level of SOD, catalase and GPx, gluthatione peroxidase, to within normal limits [27].
Green tea
Green tea extract, well known for its polyphenols, with about 40% catechins, has been suggested to be useful in the treatment of androgen-mediated skin disorders including acne [28]. Green tea exerts its effect through mechanisms, including antibacterial effect through anti-adhesive activity against P. acnes and staph aureus [29]. Green tea has been shown to impede local and systemic declines in SOD and GSH-Px activity whereby attenuates lipid peroxidation [30].
Nicotinamide
Nicotinamide, essentially an antioxidant, exerting most its effects via poly-adenosine diphosphate-ribose polymerase inhibition shows comedolytic activity [31]. 4% nicotinamide gel is of comparable efficacy to 1% clindamycin gel in the treatment of acne vulgaris by providing potent antiinflammatory activity without the risk of inducing bacterial resistance [32]. Systemic use of nicotinamide for inflammatory acne has also been shown effective after 2 months of use [33].
Human studies have demonstrated that lutein and zeaxanthin are present in the skin, and animal studies have provided evidence of significant efficacy against light-induced skin damage, especially the ultraviolet wavelenghts [34]. Topical application of lutein has been demonstrated to decrease up to 60% in lipid peroxidation and its use in evidence based skin care for maintenance of acne management is warranted [34].
Lactoferrin
lactoferrin is a milk protein with inhibitory effect on lipid peroxidation induced by oxidative stress [35]. Lactoferrin-enriched fermented mild ameliorates acne lesions with a selective decrease of triacylglycerols in skin surface lipids [36].
Vitamin E
An open label study over an eight-week period demonstrated clinical efficacy of application of topical vitamin E in acne patients of mild to moderate severity. This study which was to evaluate limiting contribution of free radical oxidation through using vitamin E for acne holds a sufficient promise as an impetus for future studies. The skin cleaves to a stratagem to offset squalene-by-products maleficence by provision of tocopherols in the sebum. Sebum of acne patients was found having lower level of vitamin E, α and γ tocopherols, and higher in polar lipids, in large, lipid peroxidation metabolites compare to that of control.
An anti inflammatory polyphenol (hydroxystilbene) from grapes, also found copiously in knotweed (Polygonum cuspidatum), which downregulates K13/mTORC1 (lipid kinases) signaling leading to an inhibitory impact on lipogenesis in sebocytes. Resveratrol has also been demonstrated to possess anti bacterial properties and can impede proliferation of C. Acnes. Another comparative study surveyed antibacterial effect of benzoyl peroxide against resveratrol finding the first with bactericidal effect vanished in 24 hours while the later carrying tenacious bacteriostatic properties leading to bacterial cell death over a period of several days.
It is of note that the word non inflammatory acne is recognized as a misnomer by most recent research as acne precursors and comdedones carry some level of inflammation so the previous classification as inflammatory and non inflammatory acne has been suggested to be discontinued.
One study posits successful treatment of twenty patients with inflammatory acne vulgaris after using resveratrol for sixty days in a half-face study assessing the results by global acne grading system (GAGS). This single-blind study resulted in 66.7% improvement in total micocomedones in resveratrol treated side of the face. A double blind randomized parallel group study on 100 women to examine protective effect of polyphenols against oxidative stress on various skin parameters including sebum production indicated that only two weeks after oral consumption of Olea europaea leaf, Lippia citriodora, Rosmarinus officinalis, and Sophora japonica skin elasticity, depth of wrinkles and skin lipogenesis meaningfully improved. Mean diminished sebum production was 3.9 (P = 0.0010), 5.1 (P = 0.0001), 9.5 (P < 0.0001) and 10.9% (P < 0.0001) after 2, 4, 8 and 12 weeks of treatment, respectively.

The complex pathogenesis of acne vulgaris extends far beyond the simplistic binary of bacterial colonization and hormonal stimulation, with emerging evidence pointing to lipid peroxidation as a critical, yet underappreciated, driver of inflammation and lesion formation. The peroxidation of squalene—an abundant, highly unsaturated lipid in human sebum—triggers a cascade of oxidative stress, cytokine production, and subsequent inflammatory responses that contribute significantly to acne pathophysiology. Squalene peroxides not only initiate comedogenesis but also deplete intrinsic skin antioxidants such as glutathione, thereby exacerbating oxidative damage. This oxidative stress perpetuates a self-reinforcing inflammatory cycle, as ROS generation further depletes antioxidant defense mechanisms, including glutathione peroxidase (GSH-Px) and superoxide dismutase (SOD), which have been consistently reported to be lower in acne patients.
Importantly, these oxidative-inflammatory mechanisms are also implicated in adult acne, a condition often neglected in dermatologic discourse but increasingly prevalent, especially among women. Given the overlap between oxidative stress pathways involved in aging and acne, a more integrative therapeutic strategy that addresses both skin aging and acne-associated inflammation is warranted.
TLR-2 signaling and NADPH oxidase activation by Cutibacterium acnes (formerly Propionibacterium acnes) within keratinocytes further compound the inflammatory response by stimulating IL-8 production and triggering MAPK pathways. The inflammatory phenotype of acne is not unique to acne vulgaris but is also evident in acne inversa and hidradenitis suppurativa, both of which demonstrate shared dietary and immunological triggers, emphasizing the systemic nature of inflammatory acne diseases.
Although conventional treatments, including antibiotics and retinoids, are widely prescribed, their mechanisms may be partly attributable to antioxidant properties, rather than solely antimicrobial or comedolytic effects. Tetracyclines like minocycline, for instance, exert notable free radical scavenging capabilities, reducing superoxide and IL-8 production independent of bacterial suppression. However, the development of antibiotic resistance and safety concerns, particularly during pregnancy, challenge their long-term utility.
This evolving understanding of acne’s oxidative foundation has prompted increasing interest in non-pharmacologic and antioxidant-based therapies. A diverse array of antioxidants—including sodium L-ascorbyl phosphate (SAP), zinc ascorbate, green tea polyphenols, resveratrol, nicotinamide, lutein, lactoferrin, and vitamin E—has demonstrated efficacy in reducing lipid peroxidation, modulating sebogenesis, and dampening inflammatory responses. SAP, in particular, has shown synergistic potential with retinol and even superior outcomes compared to clindamycin in clinical trials. Similarly, zinc ascorbate mimics SOD activity while exerting potent antimicrobial effects beyond its constituent compounds. These agents not only curb ROS formation but, in many cases, modulate hormone-mediated pathways, such as DHT suppression via DKK-1 regulation, further reinforcing their relevance in acne management.
Green tea extract, nicotinamide, and resveratrol stand out for their multifaceted mechanisms: reducing bacterial adherence, preserving antioxidant enzyme activity, and interfering with lipogenesis signaling (e.g., mTORC1), respectively. In particular, resveratrol’s prolonged bacteriostatic activity against C. acnes, combined with its anti-inflammatory and sebostatic effects, marks it as a promising natural agent. Studies using oral polyphenols have further validated the systemic benefits of antioxidants, including reductions in sebum production and improved skin parameters within weeks of administration.
Given these findings, the categorization of acne into inflammatory and “non-inflammatory” types is increasingly viewed as obsolete. Even microcomedones exhibit inflammatory features at the molecular level, suggesting that oxidative stress and inflammation are universal across all acne lesions. This underscores the need to revise current treatment paradigms and incorporate lipid peroxidation and oxidative balance as central therapeutic targets.
In conclusion, the role of lipid peroxidation in acne vulgaris provides a compelling framework for reimagining treatment strategies, particularly through the lens of antioxidant therapy as a non-prescription, evidence-based alternative. By disrupting the oxidative-inflammation cycle and restoring skin’s redox balance, such approaches may offer safer, more sustainable, and multi-beneficial interventions for individuals affected by acne, especially those seeking alternatives to antibiotics and retinoids. Future clinical research should aim to validate these interventions through larger, well-controlled studies, and further elucidate the biochemical mechanisms underpinning antioxidant efficacy in acne-prone skin.
1. Zouboulis CC. Acne and sebaceous gland function. Clin Dermatol. 2004;22(5):360-6.
2. Lorincz AL. Human skin lipids and their relation to skin diseases. 1965:1-12.
3. Tappel AL. In: lipid peroxidation and fluorescent molecular damage to membranes, pathological aspects of cell membranes. Trump DF, Arstila A. 1974.
4. Bowe WP, Logan AC. Clinical implications of lipid peroxidation in acne vulgaris: old wine in new bottles. Lipids Health Dis. 2010;9:141.
5. Mills OH, Porte M, Kligman AM. enhancement of comedogenic substances by ultraviolet radiation. Br J Dermatol. 1978;98:145-50.
6. Ottaviani M, Alestas T, Zouboulis CC, Picardo M. Peroxidated squalene induces the production of inflmmatory mediators in HaCaT keratinocytes: a possible role in acne vulgaris. Invest Dermatol 2006;126:2430-7.
7. Chiba K, Yoshizawa K, Makino I, Kawakami K, Onoue M. changes in the levels of glutathione ater cellular and cutaneous damage induced by squalene monohydroperoxide. J Biochem Mol Toxicol. 2001;15:150-8.
8.Tochio T, Tanaka H, Nakata S, Ideno H. Accumulation of lipid peroxide in the content of comedones may be involved in the progression of comedogenesis and inflammatory changes in comedones. J Cosmet Dermatol. 2009;8:152-8.
9. Portugal M, Barak V, Gisnburg I, Kohen R. Interplay among oxidants, antioxidants and cytokines in skin disorders: presest status and future considerations. Biomed Pharmacother. 2007;61:412-22.
10. Burkhart CN, Gottwald L. Assessment of etiologic agent in acne pathogenesis. Skinmed. 2003;2(4):222-8.
11. Grange PA, Chereau C, Raingeaud J, Nicco C, Weill B, Dupin N, Batteux F. Production of superoxide anions by keratinocytes initiates P. acnes-induced inflammation of the skin. PLoS Pathog. 2009;5:e1000527.
12. Basak PY, Glutekin F, Kilinc I. The role of the antioxidative defense system in papulopustular acne. J Dermatol. 2001;28:123-7.
12-1. Michaelsson G et al. Erythrocyte glutathione peroxidase activity in acne vulgaris and the effect of selenium and vitamin E treatment. Acta Derm Venerol 1984; 64:9-14.
12-2. Zouboulis CC. Is acne vulgarism a genuine inflammatory disease? Dermatology, 2001; 203(4): 277-9.13. Sarici G, Cinar S, Armutcu F, Altinyazar C, Koca R, Tekin NS. Oxidative stress in acne vulgaris. J Eur Acad Dermatol Venereol. 2010;24:763-7.
12-3. Farrar MD et al. Acne: inflammation. Clin Dermatol, 2004; 22(5): 380-4.
13. Sarici G, Cinar S, Armutcu F, Altinyazar C, Koca R, Tekin NS. Oxidative stress in acne vulgaris. J Eur Acad Dermatol Venereol. 2010;24:763-7.
14. Antioxidant properties of minocycline: neuroprotection in an oxidative stress assay and direct radical-scavanging activity.
14-1 Biswal I, et al. In vitro antimicorbial susceptibility patterns of Propionibacterium acnes isolated from patients with acne vulgaris. J Infect Dev Ctries 2016;10(10):1140-1145
14-2 Bayerl C. Acne therapy in pregnancy. Hautarzt 2-13;64(4):269-73
14-3. Nachos K et al. Propionibacterium acnes is developing gradual increase in resistance to oral tetracyclines. J Med Microbial 2017; 66(1):8-12.
15. Subantimicorbial-dose doxycycline in the treatment of moderate facial acne. J Drugs Dermatol. 2008;7:1149-52.
16. Harder J, Schroder J-M, Glaser R. The skin surface as antimicrobial barrier: present concepts and future outlooks. Exp Dermatol. DOI: .
17. Grice E A, Segre J A. The skin microbiome. Nat Rev Micoriol. 2011;9:244-53.
17-1. Chehoud C et al. Complement modulate the cutaneous microbiome and inflammatory milieu. Proc Natl Acad Sci USA 2013 Sep 10;110(37):15061-6
18. Akamatsu H, Horia T, Hattori K. Increased hydrogen peroxide generation by neutrophils from patients with acne inflammation. Int J Dermatol. 2003;42:366-69.
18-1 Perisho K, et al. Fatty acids of acylcermides from comedones and from the skin surface of acne patients and control subjects. J Invest Dermatol 1988;90(3):350-3.
18-2. Akamatsu H, et al. Suppressive effects of linoleic acid on neutrophil oxygen metabolism and phagocytosis. J Invest Dermatol 1990;95(3):271-4.
18-3. Puhvel SM et al. The chemoattractant properties of comedonal components. J Invest Dermatol 1978;71(5):324-9.
19. Pinnel S, Madey D. The benefits of topical vitamin C (L-ascorbic acid) for skin care and UV-protection. J Appl Cosmetol. 1999;18:126-34.
20. Woolery-Lloyd H. Baumann L, Ikeno H. Sodium L-ascorbyl-2-phosphate 5% lotion for the treatment of acne vulgaris: a randomized, double-blind, controlled trial. J Cosmet Dermatol. 2010;9(1):22-7.
20-1. Camera E et al. Comprehensive analysis of the major lipid classes in sebum by rapid resolution high-performance liquid chromatography and electrospray mass spectrometry. J Lipid Res 2010; 51(11):3377-3788.
21. Ruamark C, Lourith N, Natakankitkul S. Comparison of clinical efficacies of sodium ascorbyl phosphate, retinol and their combination in inflammatory acne treatment. Int Cosmet Sci. 2009;31(1):41-6.
22. Klock J, Ikeno H, ohmori K, Nishikawa T, Volhardt J, Schehlmann V. Sodium ascorbyl phosphate show in vitro and in vivo efficacy in the prevention and treatment of ance vulgaris. Int J Cosmet Sci. 2005;27(3):171-6.
23. Ikeno H, Nishikawa T. An open study comparing efficacy of 5% sodium L-ascorbyl 2-phosphate lotion with 1% clindamycin gel in the treatment of facial acne vulgaris. J Am Dermatol. 2008;58(2 Suppl):AB2.
24. Zouboulis CC. The sebaceous gland. Hautarzt. 2010;61(6):476-7.
25. Kwack MH, Kim MK, Kim JC, Sung YK. L-ascorbic acid 2-phosphate represses the dihydrotestoerone-induced dickkopt-1 expression in human balding dermal papilla cells. Exp Dermatol. 2010;19(12):1110-2.
26. Linuma K, Tsuboi I. zinc ascorbate has superoxide dismutase like activity and invitro antimicrobial activity against staphylococcus aureus and Escherichia coli. Clin Cosmet Investig Dermatol. 2012;5:135-40.
27. Pathak A, Mahmood A, Pathak R, Dhawan D. Effect of zinc on hepatic lipid peroxidation and antioxidative enzymes in ethanol-fed rats. J Appl Toxicol. 2002;22(3):207-10.
28. Katiyar SK. Skin photoprotection by green tea: antioxidant and immunomodulatory effects. Curr Drug Targets Immune Endocr Metabol Disord. 2003 Sep;3(3):234-42.
28-1. Vayalil PK et al. Treatment of green tea polyphenols in hydrophilic cream prevents UVB-induced oxidation of lipids and proteins, depletion of antioxidant enzymes and phosphorylation MAPK proteins in SKH-1 hairless mouse skin. Carcinogenesis. 2003;3(3):234-42
29. Lee JH, Shim JS, Chung MS, et al. In vitro anti-adhesive activity of green tea extract against pathogen adhesion. Phytother Res. 2009;23(4):460-6.
30. Xu Y, Zhang JJ, Liu H. Green tea polyphenols inhibit cognitive impairment induced by chronic cerebral hypoperfusion via modulating oxidative stress. J Nutr Biochem. 2010;21:741-8.
31. Otte N, Borelli C, Korting HC. Nicotinamide biologic actions of an emerging cosmetic ingredient. Int J Cosmet Sci. 2005;27:255-61.
32. Shalita AR, Smith JG, Parish LC, Sofman MS, Chalker DK. Topical nicotinamide compared with clindamycin gel in the treatment of acne vulgaris. Int J Dermatol. 1995;34:434-7.
33. Niren NM, Torok HM. The nicomide improvement in clinical outcomes study: results of an 8-week trial. Cutis. 2006;77(1 Suppl):17-28.
34. Roberts R L, Green J, Lewis B. Lutein and zeaxanthin in eye and skin health. Clin Dermatol. 2009;27(2):195-201.
34-1. Gonzalez S et al. Dietary lutein/zeaxanthin decreases ultarviolet B-induced epidermal hyperproliferatio and acute inflammation in hairless mice. J Invest Dermatol. 2003;12(12):399-405.
34-2. Pentland AP et al. Bradykinin-induced prostaglandin synthesis is enhnaced in keratinocytes and fibroblasts by UV injury. Am J Physiol. 1991;261(3Pt2):R543-7.
34-3. Odabasoglu F et al. α-Lipoic acid has anti-inflammatory and anti-oxidative properties: an experimental study in rats with carrageenan-induced acute and cotton pellet-induced chronic inflammations. Br J Nutr. 2011; 105(1):31-43.
34-4. Khalili M et al. Does lipoic acid consumption affect the cytokine profile in multiple sclerosis patients: a double-blind, placebo-controlled, randomized clinical trial. Neuroimmunomodulation. 2014; 21(6):291-6.
34-5. Spain R et al. Lipoic acid in secondary progressive MS: A randomized controlled pilot trial. Neurol Neuroimmunol Neuroinflamm. 2017 Sep; 4(5):e374.
34-6. Venkatraman MS et al. Alpha lipoic acid-based PPARgamma agonists for treating inflammatory skin disease. Arch Dermatol Res 2004;296(3):97-104.
34-7. Yildirim Bas F et al. Effect of alpha lipoic acid on smoking-indduced skin damage.Cutan Ocul Toxicol 2017;36(1):67-73.
34-8. Fasano E et al. antioxidant and anti inflammatory effects of selected natural compounds contained in a dietary supplement on two immortalized keratinocyte lines. Biomed Res Int 2014;2014:327452.
35. Konishi M, Iwasa M, Yamauchi K, et al. Lactoferrin inhibits lipid peroxidation in patients with chronich hepatitis C. Hepatol Red. 2006;36:471-7.
35-1. Haversen L et al. Lactoferrin down-regulates the LPS-induced cytokine production in monocytic cells via NF-kappa B. Cell Immunol 2002;220(2):83-95.
36. Kim J, Ko Y, Cho Y. Dietary effect of lactoferrin-enriched fermented mild on skin surface lipid and clincal improvement of acne vulgaris. Nutrition. 2010;26:902-9. es in ethanol-fed rats. J App Toxicol. 2002;22(3):207-210.
37. Cunliffe WJ, Holland KT, Bojar R, et al. A randomized, double-blind comparison of a clindamycin phospate/benzoyl peroxide gel formulatio and a mtaching clindamycin gel with respect to microbiologic activity and clinical efficacy in the topical treatment of acne vulgaris. Clin Ther. 2002;24(7):1117-33.
38. Humphrey S. Antibiotic resistance in acne treatment. Skin Therapy Lett. 2012;17(9)1-3.
39. Saint-leger D. A possible role for squalene in pathogenesis of acne. In vitro study of squalene oxidation. Br J Dermatol 1986 May; 114(5)535-42.
40. Tochio T. Accumulation of lipid peroxide in the content of comedones may be involved in the progression of comedogenesis and inflammatory changes in comedones. J Cosmet Dermatol 2009 June; 8(2)152-8
41. W J Lee. Magnesium ascorbyl phosphate regulates expression of inflammatory biomarkers in cultured sebocytes. Ann Dermatol 2015 Aug; 27(4)376-82.
42. A H T Jeremy. Inflammatory events are involved in acne lesion initiation. J Invest Dermatol. 2003 July; 12(1)20-7.
43. Krautheim A. Acne: topical treatment. Clin Dermatol 2004 Sep-Oct; 22(5)398-407.
44. Ayres S Jr. Acne vulgaris and lipid peroxidation, new concepts in pathogenesis and treatment. Int J Dermatol 1978 May 17(4)305-7.
45. D T Downing. Essential fatty acids and acne. J Am Acad Derm 1986 Feb;14(2 Pt 1)221-5.
46. Selway J L. Toll like receptor 2 activation and comedogenesis, implications for pathogenesis of acne. BMC Dermatol 2013Sep 6;13:10.
47. Elias P M. The permeability barrier in essential fatty acid deficiency: evidence for a direct role for linoleic acid in barrier function