

Skin healing and scar tissue generation is a complex process entails a number of overlapping phases, including inflammation, epithelialization, angiogenesis and collagen deposition which explains multiple stages of acne scars pathogenesis suggested by Dr. Krisner from department of dermatology of university of Miami [1]. Ultimately these processes are resolved leading to a mature wound and macroscopic production of scars as a result of prolonged acne. This process occurs in formation of scarring as most resolving acne lesions are associated with some degree of damage to the skin. Although inflammation and repair mostly occur concurrently, the sensitivity of the process is underscored by the consequences of disruption of the balance of regulatory cytokines.
Consequently, cytokines, which are central to this constellation of events, have become targets for therapeutic intervention to modulate the wound healing process [2]. One example is use of nicotinamide in treatment of chronic inflammatory acne by blocking IL8 causing downregulation of P.acnes -induced IL-8 promoter activation [3]. Depending on the cytokine and its role, it may be appropriate to either enhance (recombinant cytokine, gene transfer) or inhibit (cytokine or receptor antibodies, soluble receptors, signal transduction inhibitors, antisense) the cytokine to achieve the desired outcome [4]. Comparison of scar-free wound healing in embryo to adult scars signifies role of transforming growth factor-beta, (TGF-beta) family in controlling the outcome [5]. TGF-beta3 elicits a scar-free healing regenerative healing response, whereas TGF-beta1 and TGF-beta2 elicit a fibrotic scarring response [5].
Response to injury in the skin initiated by coagulation and an acute local inflammation. This follows by mesenchymal cell migration, proliferation and matrix synthesis. Failure to resolve the inflammation can lead to chronic nonhealing wounds, whereas uncontrolled matrix accumulation, often involving aberrant cytokine pathways, leads to excess scarring and fibrotic sequelae. Reduced and more transient expression of TGF-beta and their receptors was observed in non fetal wounds compared with adult wounds [6]. Manipulation of cytokines using exogenous growth factors provides therapeutic opportunities to control abnormal wound healing and acne scar formation [7].
Connective tissue repair and acne scars pathogenesis: where repair cannot be accomplished with resolution, scar tissue forms. One challenging question here would be why acne sequels with atrophy and not hypertrophy is most commonly seen. One hypothesis is that an imbalance in the ratio of matrix metaloproteinases to tissue inhibitors of metaloproteinases results in the development of trophic or hypertrophic scars. Groupe experts acne proposed that inadequate response results in diminished deposition of collagen factors and formation of an atrophic scar while, if the healing response is too exuberant, a raised nodule of fibrotic tissue forms hypertrophic scars [8].
Fibroblasts migrate to the site of injury and proliferate following stimulation by TGF-beta and many other cytokines and growth factors. The presence of TGF-beta in the granulation tissue was expected to be important for efficient healing, since TGF-beta was shown to stimulate angiogenesis, fibroblast proliferation, myofibroblast differentiation and matirix deposition [9]. Their biological effects are mediated by heteromeric recepotor complexes, which activate intracellular signaling cascades [10]. Immediately after wounding, TGF-beta1 is released in large amounts from platelets [11]. which in turn causes chemotaxis for neutrophils, macrophages and fibroblasts. These cells further release TGF-betas. This includes all isofroms of TGF-beta that are in demand during the whole process of wound healing [12]. The presence of TGF-beta in the granulation tissue was expected to be important for efficient healing, since TGF-beta was shown to stimulate angiogenesis, fibroblast proliferation, myofibroblast differentiation and matrix deposition [13]. Aberrant expression of TGF-beta is associated with wound healing defect seen in glucocorticoid treated mice [14].
New endothelial cells proliferate upon induction by VEGF. This forms granulation tissue within 3-5 days of injury. Granulation tissue is pink in appearance and characterized by the formation of new vessels, production of ECM, fibrosis, and initial repair that lays down the structure on which the final scar will be formed. Angiogenesis proceeds by generation of new leaky vessels from existing vessels [15]. Basic fibroblast growth factor and VEGF induce angiogenesis [16]. Fibrosis develops with deposition of ECM by fibroblasts. Collagen synthesis and decreased degradation of collagen gives strength to the healing wound. IL-1, TNF, TGF-beta, b-FGF all work to increase the production of collagen locally. Keratinocytes interacts with fibroblasts in a TGF-beta dependent manner during the wound healing process [17].
Re-epithelialization and acne scars pathogenesis: Specialized enzymes released from macrophages degrade collagen (metalloproteinases, gelatinases, and collagenases) to remodel the wound and form the final scar. Keratinocytes strongly express TGF-beta1 and TGF-beta3 and waakly express TGF-beta2 [18]. TGF-beta1 modulates differentiation of fibroblasts to myofibroblasts which contribute to generation of a contractile force [19].
Skin injury, blood vessel damage, coagulation response, platelet aggregation, release of cytokines from aggregated, degranulating platelets, TGF-beta (transforming growth factor), PDGF (platelet derived growth factor), VEGF (vascular endothelial growth factor), leakage of plasma proteins influenced by VEGF, involvement of TGF-beta in all steps of wound healing from the initial clot generation to matrix formation and remodeling. Of much importance, is aggregation of platellets to themselves and to exposed collagen, releasing TGF-beta which, in turn, initiates the repair process [20].
Release of adhesion molecules, (VCAM, ICAM, ELAM) induced by cytokines, facilitated extravasation of leukocytes by adhesion molecules, release of proteases from leukocytes mediated by TGF-beta, IL-1, TNF. This causes phagocytosis of debris, microbes and degraded matrix components. Neutrophil recruitment typically peaks around 24-48 hours post wounding, followed by an increasing representation of monocytes which are essential for optimal wound healing and explains a component of events in acne scars pathogenesis.
By examining biopsy specimens of inflammatory acne lesions from the back of patients with severe acne scars and without scars to investigate acne scars pathogenesis, Holland et al. found that the inflammatory reaction at the pilosebaceious gland was stronger and had a longer duration in patients with scars versus those without, in addition, the inflammatory reaction was slower in those with acne scars versus patients who did not develop scars [21]. they showed a strong relationship between severity and duration of inflammation and the development of scars, suggesting that treating early inflammation in acne lesions may be the best approach to prevent acne scars [22].
Resolution of a wound of infection agents and debris triggers relief of inflammation and followed by migration and proliferation of epithelial cells. This ensures closure of exposed area of the wound. Migration of epithelial cells is dependent to the growth factors and loss of contact between adjacent cells. Once contact is established between opposing keratinocytes, migration stops. This is mainly mediated by TGF-beta [23].
Underlying this newly-formed epithelium, granulation tissue forms. Granulation tissue consists of fibroblasts, which deposit collagen, and new vessels [13]. Retinoids downregulate the tensile tension of fibroblasts in collagen matrices by the induction of tenascin-C [24]. Neovascularization is under the control of different cytokines. TNF, IL-1 and TGF-beat are among the important ones. This process is under a tight control and there are cytokines responsible to stop the process at certain points.
The initial stages of angiogenesis wound healing is induced by a non-angiogenic process involving myofibroblast contraction, differentiated by help of TGF-beta, followed by sprouting angiogenesis which is regulated by different growth factors including TGF-beta [25].
By deposition of granulation tissue, fibroblast initiates collagen deposition. TGF-beta contributes to the fibrotic process by stimulating fibroblasts to synthesize collagen I, III and V, proteoglycans and fibronectin. TGF-beta also inhibits proteases, favoring matrix accumulation. In contrast, PDGF, released by activated macrophages, fibroblasts and endothelial cells, upregulates proteases. PDGF, also has a role in matrix deposition. TGF-beta3 is involved in maturation of scar tissue by regulating epidermal cell migration, collagen deposition and cellular proliferation in rabbit wound healing [26].
IL-1 and TNF also influence wound healing either directly by affecting fibroblasts or endothelial cells or indirectly by inducing other cytokines and growth factors. Exogenous addition of TGF-beta3 to cutaneous wounds in pre-clinical (adult) in vivo models reduces early extracellular matrix deposition and these molecules are deposited with a markedly improved architecture in the neodermis [27].
This phase is also cytokine mediated. Enzymes released from keratinocytes and fibroblasts including serine proteases, elastase and matrix metaloproteases function to deposit, remove and organize collagen and other matrix proteins, suggested by Dr. Jean Schwarzbauer from department of molecular biology of Princeton university [28]. Tensile strength of the scar tissue would not be as strong as the original tissue, however, it is comparable in many respects.
The more prolonged the inflammation, the more likely formation of scar tissue. Even though proteases work to eliminate excess collagen, as long as the inflammation persists, matrix deposition by TGF-beta is active. Inhibition of this growth factor is induced by various immunoglobulins and decorin which act to reduce scarring. IL-10 and inhibitors of TNF also demonstrate some activity in collagen removal and activation of proteases.
In short, transforming growth factor beta family’s contribution in various phases of wound healing and scar development is significant and explained by regulating production of extracellular matrix, proteases, protease inhibitors, chemotaxis and proliferation of stem cells which, in turn, control scar contraction, angiogenesis, granulation tissue formation and scar maturation. Therapeutic values of topical application TGF-beta1 inhibitors in hypertrophic scars with increased expression of TGF-beta1 has been studied was demonstrated by a wound healing research group in university of Edmonton [29].
Keratinocytes have been found in possession of the andronergic receptors, β2 and α, conferring a neuroendocrine cloak on epidermis and withal competent for de novo generation and degradation of catecholamines. Upon activation, these receptors illicit an inhibitory response on migration of keratinocytes from the basal epidermis to the surface of the wound, giving rise to delayed wound healing. This finding may justify how stress and its subsequent adrenaline/noreadrenlain rise may impact healing and acne scars. By the same token, timolol and other beta blockers have been employed in dermatological disease armamentarium such as infantile hemangiomas, epidermolysis bullosa, kaposi syndrome, acne, acne scars and rosacea.
The epidermis of acne scars was characterized by keratin plugging in the hair follicle orifice and multichannelled tracts [30]. The dermis of the acne scars had characteristics including a decrease in the dermal thickness and loss of pilosebaceous units and resultant decrease in skin surface lipids production. In addition, inflammatory cell infiltrates were seen in the dermis and insufficient dense collagen fiber deposition was found in the whole dermis [30][*].
Application of %95 tricholoroacetic acid (TCA) repeated at 6-week intervals for a total of six treatments revealed apparent cosmetic improvement in both depth and appearance of acne scars [31]. Patient satisfaction was high. Histologic examination demonstrated a decrease in the depth of acne scars as well as increased collagen fibers and fragmentation of elastic fibers were noted.
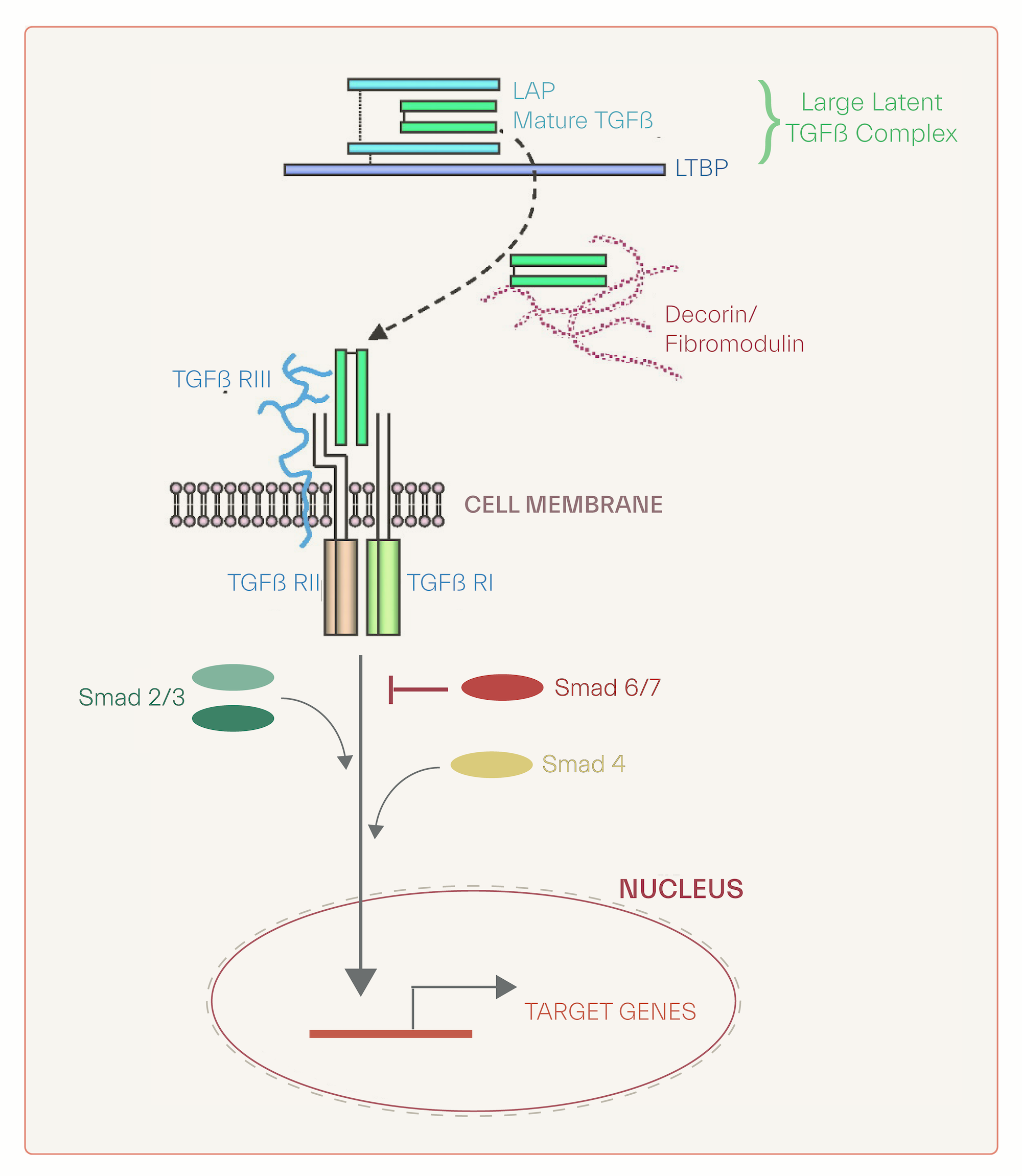
TGF-beta in its inactive form binds to latency associated protein (LAP), which, in turn, binds to TGF-Beta binding protein (LTBP) resuling in activation of TGF-beta. This activated molecule is either sequestered by extracellular binding proteins such as decorin, fibromodulin or it binds to a type III receptor that present it to a singal transducing recptors (type I and type II). This binding elicits phosphorylation of type I receptor causing binding and phosphoryaltion of smad2 and smad3. Phyosphorylated smads bind to smad4 and migrate to nucleous for binding to transcription factors and expression of target genes [7].. TGF-beta isoforms are considered of inhibitory effect on keratinocytes proliferation while exhibit distinct impact on dermal fiber-producing cells, inciting their production.
Healing by First Intention involves healing of clean edges such as a surgical cut. A fibrin clot closes the wound quickly. Neutrophils are evident within 24 hours, followed by epithelial cell migration along the cut surfaces to meet within 48 hours. Macrophages and granulation tissue predominate by day 3 with collagen fibers being secreted. Granulation tissue and angiogenesis are evident by 3-5 days. Collagen becomes organized to bridge the wound by the end of the first week. Functional epidermis is formed, although dermal structures are permanently disrupted. After one week, would strength is only 10% of normal, but increases to 70-80% by 3 months.
Healing by second Intention occurs with more serious wounds that are larger, less linear, with edges not opposed. Inflammation and scarring leads to wound contraction at the margins. Granulation tissue will cover a larger area, persist for longer, and develop into thicker scar tissue. Secondary infection and a heightened inflammatory response is more likely. Infection, corticosteroids, and poor perfusion decrease the rate of healing. Location of the injury and the type of tissue also matters.
The pathogenesis of acne scars reflects a complex interplay between inflammatory response, cytokine signaling, and the wound healing cascade, with Transforming Growth Factor-beta (TGF-β) playing a pivotal role throughout. Acne scars arise not merely from the presence of inflammation, but from the dysregulation of the resolution phase of healing. This leads to either atrophic or, less commonly, hypertrophic scarring — depending on the balance between matrix synthesis and degradation, the intensity and duration of inflammation, and individual immunological responses.
Evidence suggests that most acne scars are atrophic due to inadequate collagen deposition, driven by a reduced or transient reparative response. Conversely, hypertrophic scars result from excessive fibroblast activation and collagen accumulation, likely influenced by persistent TGF-β1 and β2 signaling. The differential effects of TGF-β isoforms (β1 and β2 promoting fibrosis vs. β3 promoting regenerative healing) underscore the therapeutic potential of isoform-specific modulation in acne scar prevention and treatment.
The transition from inflammation to tissue repair is cytokine-regulated, with TGF-β, IL-1, TNF, and VEGF acting as key mediators. Aberrations in these pathways — especially prolonged expression of pro-fibrotic cytokines — result in granulation tissue that matures into fibrotic scars rather than resolving into normal dermis. Moreover, the early inflammatory phase has been identified as critical: studies such as those by Holland et al. suggest that prolonged inflammation, particularly in the pilosebaceous unit, correlates with greater scar severity. This finding reinforces the need for early and aggressive acne treatment to interrupt inflammatory cascades before dermal remodeling becomes pathological.
Keratinocyte and fibroblast interactions, largely governed by TGF-β signaling, are also central to epithelial regeneration and dermal matrix organization. Delays in keratinocyte migration — exacerbated by β-adrenergic stimulation or systemic stress responses — may contribute to impaired re-epithelialization and pro-scarring environments. This highlights an interesting neuroimmunological angle, where stress-related hormones may directly influence scar outcome, providing rationale for beta-blocker-based topical therapies in acne scars.
Further, the histopathology of acne scars — including reduced dermal thickness, loss of pilosebaceous units, and poor collagen organization — confirms that scar formation reflects a failure of coordinated tissue regeneration, rather than merely excess inflammation. Interventions that promote organized collagen synthesis (e.g., retinoids, fractional lasers, microneedling, or high-concentration TCA) appear to restore structural integrity and improve clinical appearance, as evidenced by post-treatment histology showing increased collagen fiber density and fragmentation of abnormal elastin.
At the molecular level, TGF-β receptor signaling through Smad pathways and its regulation by binding proteins (e.g., decorin, LTBP) determines much of the fibrotic vs. regenerative outcome. Targeting this axis remains a compelling therapeutic strategy. For instance, exogenous TGF-β3 or inhibitors of TGF-β1 have shown promise in preclinical models to modulate scar formation and improve extracellular matrix architecture.
Ultimately, acne scarring must be viewed not as a singular event, but as the failure of a tightly regulated wound healing program, shaped by host immunity, inflammation control, cytokine signaling, and tissue-specific responses. Therapeutic strategies should aim not only at treating visible scars but also at modulating upstream immune and molecular pathways to prevent their formation. This includes the use of anti-inflammatory agents, growth factor modulators, and perhaps even gene or cytokine-based therapies to guide healing toward a regenerative rather than fibrotic trajectory.
1. Robert krisner, Jie Li, Juan Chen. Pathophysiology of acute wound healing. Clin Dermatol. 2006;25(1):9-18.
2. Wolfram D, Tzankov A, Pulzl P, Piza-katzer H.
Hypertrophic scars and keloids: a review of their pathophysiology, risk factors and therapeutic management. Dermatol Surg. 2009;35(2):171-81.
3. Grange PA, Raingeaud J, Calveez V, Dupin N.
Nicotinamide inhibits propionibacterium acnes-induced IL-8 production in kderatinocytes through the NF-kappaB and MAPK pathways. J Dermatol Sci. 2009;56(2):106-12.
4. Martin P, Leibovich SJ. Inflammatory cells during wound healing. Trends Cell Biol. 2005;5(11):599-607.
5. Ferguson MW, OKane S.
Scar-free healing: from embryonic mechanisms to adult therapeutic intervention. Philos Trans R Soc B Biol Sci. 2004;359(1445):839-50.
6. Cowin AJ, Holmes TM, Brosnan P, Fergusan MW. Expression of TGF-beta and its receptors in murine fetal and adult thermal wounds. Eur J Dermatol. 2001;11:424-31.
7. Werner S, Grose R. Regulation of wound healing by growth factors an
d cytokines. Physiol Rev. 2003;83(3):835-70.
8. Chivot M, Pawin H, Beylot C, et al. Acne scars epidemiology, physiopathology, clinical features and treatment. Ann Dermatol Venereol. 2006;133(10):813-24.
9. Roberts AB, Sporn MB. Transforming growth factor-beta. The Molecular and cellular biology of wound repair. 1996, 275-308.
10. Menke N, Deigelmann RF.
Biochemical pathways of wound healing: Implications for development of disease-specific diagnostics. Advances in Clinical Chemistry. 2006;41:167-87.
11. Soron MB, Komoriya A, Meyers CA, Miller DM.
Transforming growth factor-beta in human platelets. Identification of a major storage site, purification and characterization. J Biol chem. 258:7155-7160, 1983.
12. Roberts AB, Sporn MB. Transforming growth factor-beta. In: The molecular and cellular biolgoy of wound repair(2nd edition), edited by Clark RAF. New York:Plenum, 1996, p. 275-308.
13. Desmouliere A, Gabbiani G.
Transforming growth factor-beta1 induces alpha smooth muscle actin expression in granulation tissue myofibroblasts and in quiescent and growing cultured fibroblasts. J Cell Biol. 1993;122:103-111.
14. Madlener M, Frank S, Werner S.
Transforming growth factors beta1, beta2, beta3 and their recepotors are differentially regulated during normal and impaired wound healing. J Biol Chem. 1996;271:10188-10193.
15. Bicknell R, Harris AL. Novel angiogenic signaling pathways and vascular targets. Annu Rev Pharmacol Toxicol. 2004;44:219-238.
16. Gurtner GC, Werner S, Barrandon Y, Longaker MT. Wound repair and regeneration. Nature. 2008;453:314-321.
17. Werner S, Krieg T, Smola H. Keratinocyte-fibroblast interaction in wound healing. J Invest Dermatol. 2007;127:998-1008.
18. Edward M, et al. Keratinocyte reuglation of TGF-beta and connective tissue growth factor expression: a role in suppression of scar tissue formatio. Wound Repair Regen. 2007;15:748-55.
19. Harvey KA, et al.
Diverse signaling pathways regulate fibroblast differentiation and transformation through Rho kinase activation. J Cell Physiol. 2007;211:353-63.
20. Wakefield LM, Roberts AB. TGF-beta singlaing: Positive and negative effects on tumorigenesis. Curr Opin Genet Dev. 2002;1:22-29.
21. Fabbrocini G, Monfrecola G, et al. Acne scars: pathology, classifications and treatment. Dermatol Res Pract. 2010;893080.
22. Cunliffe WJ, Holland DB, Layton AM, et al.
Inflammation in acne scarring: a comparison of the responses in lesions from patients prone and not prone to scar. Br J Dermatol. 2004;150(1):72-81.
23. Stadelmann WK, Digenis AG, Tobin GR. Physiology and healing dynamics of chronic cutaneous wounds. Am J Surg. 1998;176(2A Suppl):26S-38S.
24. Yamada E, Shimizu M, et al.
Tocoretinate inhibited the contractionof collagen gel matrices by human dermal fibroblasts with tenascin-c expression. J Dermatol Sci. 1999;22(1):45-53.
25. Kilarski WW, Samolov B, Petersson L, Kvanta A, Gerwins P. Biomechanical regualtion of blood vessel growth during tissue vascularization. 2009;15:657-64.
26. Woodley DT, Li W et al.
A traffic control role for TFG-beta3 orchestrating dermal and epidermal cell motility during wound healing. J Cell Biol. 2006;172:1093-1105.
27. Occleston NL, Laverty HG, Ferguson MW.
Prevention and reducation of scarring in the skin by transforming growth factor beta 3: from laboratory discovery to clinical pharmaceutical. J Biomater Sci Polym Ed;200819(8):1047-63.
28. Midwood KS, Williams LV, Schwarzbauer JE. Tissue repair and dyanmics of the extracellular matrix. Int J Biochem Cell Bio. 2004;36(6)1031-1037.
29. Tradget EE, Wang JF, Jiao H, Shankowsky HA, et al. Fibrocytes from burn patients regulate the activities of fibroblasts. Wound Repair Regen. 2007;15:113-121.
30. Lee WJ, Jung HJ, et al.
Serial sections of atrophic acne scars help in the interpretation of microscopic findings and the selection of good therapeutic modalities. J Eur Acad Dermatol Venereol. 2011;10:1468-3083.
31. Howard MS. Kent DE, Yug A, Lane JE.
Histologic study of depressed acne scars treated with serial high concentration (95%) tricholoroacetic acid. Dermatol Sur. 2006;32(8):985-90.
* Connoly D. Acne scarring, pathogensis, evaluation and treatment options. J Clin Aesthet Dermatol, 2017 Sep; 10(9):12-23